|
A series of dengue vaccines are available, however, only a few are effective. The first dose of the vaccine protects against a single dengue infection, and this protective effect can last for years. However, it is possible to develop vaccine-enhanced disease if patients are not immune to the dengue virus. The global incidence of dengue is rising. Vaccines are the only way to prevent a dengue infection from becoming a major issue for communities in the developing world.
The development of dengue vaccines is currently in its nascent stage, however, the growing incidence of dengue fever in Asia is creating a lucrative opportunity for pharmaceutical companies. For example, Takeda Pharmaceuticals, a leading Japanese pharmaceutical company, has recently begun regulatory submissions for its dengue vaccine candidate, TAK-003, in the EU and in dengue-endemic countries. With the help of these partnerships, the development of dengue vaccines is moving forward. The Global Dengue Vaccine Market is estimated to account for US$ 1,262.0 Mn in terms of value by the end of 2027. While the results of this study were underwhelming, the vaccine demonstrated its safety in over four thousand volunteers. There are also ongoing phase 3 studies in over thirty thousand volunteers in ten countries throughout Asia and the Americas. Further development of dengue vaccines will depend on how well they protect the population from the disease. The vaccine candidate must also be compatible with vector control measures. And, as with all vaccines, there is no guarantee of their effectiveness. However, the development of dengue vaccines has reached an important stage in its lifecycle. The availability of alternative dengue vaccines is likely to break the monopoly of Denvaxia(r) vaccine. And since the growth of the vaccine industry is likely to be concentrated in Asia-Pacific, the development of new vaccines may take years. The future of the dengue vaccine industry looks bright! And, with these developments, there are numerous new vaccines on the horizon. Sanofi Pasteur has announced that it has performed additional studies of dengue vaccines. The new studies are aimed at better describing the benefit-risk profile of dengue vaccination in seronegative individuals. The results of these studies used the newly developed NS1-based assay, and blood samples were collected 13 months after the initial vaccination. These data are useful for children aged 9 and older, those with a history of dengue, and those who live in endemic areas. Sequential vaccination has also shown promising results in the prevention of dengue. Sequential vaccination is likely to become increasingly popular as more countries adopt it as a preventative measure. As a result, firms in the global dengue vaccines industry will likely partner with researchers from the Singapore-MIT Alliance for Research and Technology. These researchers are actively seeking to develop a dengue vaccine that is both safer and more effective than existing products. While the development of dengue vaccines has been ongoing since the 1940s, progress has recently accelerated. To create a vaccine, the vaccine must balance the immune response of humans with attenuation of the virus. Moreover, the vaccine must provide complete, long-lasting protection against four serotypes of dengue while preventing the enhancement of the disease following subsequent infections. There are many challenges in developing a dengue vaccine, including the lack of an animal model for testing and the lack of basic knowledge of the disease's pathogenesis.
0 Comments
The electronic aspirin system is a novel technology designed to target the SPG nerve bundle in patients suffering from cluster headache and migraine. Moreover, the device contains a hand-held remote that blocks pain when a cluster headache begins. This device is under clinical investigation by Autonomic Technologies, Inc. The company has received authorization from the FDA to conduct clinical trials and the European regulatory authorities to begin its application in the EU. The company believes that its electronic aspirin system will effectively treat cluster headache and improve patients' quality of life.
While there are certain risks associated with the use of Electronic Aspirin, the risk of severe side effects is minimal. Most people who suffer from cluster headaches have experienced some form of pain during their headaches, and utilizing this technology has improved their quality of life significantly. A medical technologist must monitor the device to ensure its safety and to avoid any problems. There are no adverse effects reported from the use of this technology, however, some individuals may experience a decreased level of blood pressure, throat numbness, and nasal bleeding. According to Coherent Market insights the Electronic Aspirin Market Global Industry Insights, Trends, Outlook, and Opportunity Analysis, 2022-2028 The Electronic Aspirin device is a revolutionary medical technology that can significantly relieve the effects of migraine, cluster, and facial pain. Moreover, electronic Aspirin has the potential to be the preferred choice for migraine patients and those who suffer from headaches on a regular basis. These benefits may be limited to certain countries, however, the device could be widely accepted in developing countries. Analysts believe that the market for Electronic Aspirin will be steadily increasing during the forecast period. The demand for this technology will be highest in Europe, North America, and Asia Pacific. The adoption of this technology will boost its growth in the near future. Its main drivers include rising health care expenditure, technological innovations, and accelerated economic growth. The major challenges include high costs and regulatory hurdles. Although the technology is relatively new, its potential for improving neurological disorders is extremely high. With the ability to treat cluster headaches, it may even prove to be effective in the treatment of other pain-related disorders. Headaches are a common complaint among adults. The World Health Organization reports that around half of the population worldwide has some form of headache disorder. Approximately one billion people suffer from migraine. A miniaturized implantable neurostimulation device from ATI has been approved for migraine and cluster headache pain relief. The company's Pulsante SPG Microstimulator system has an expanded indication in Europe. It is the first implantable neurostimulation therapy to receive a CE mark. Its CE mark confirms the device's efficacy. Electronic Aspirin is intended to block pain in cluster headache sufferers and improve their quality of life. In recent years, medical technology has made great advances in the field of pain management. Along with easing headaches, electronic aspirin has been developed to treat other types of neurological disorders, including cluster headaches. This new technology is implanted into the upper gum, where it can be positioned permanently for a lifetime. Its design enables the device to be inserted without the need for a painful procedure. It is an excellent medical innovation, however, it should be used only by physicians. CAR T Cell Therapy is a new and promising treatment for blood cancers development. It uses genetic engineering to create artificial T cell receptors. These cells are then used in immunotherapy.
This FDA has approved this treatment for curing certain blood cancers. CAR T cell therapy has shown great promise in treating many types of blood cancers. A new study by Stanford University suggests that more than half of CAR-T cell therapy patients have relapsed, the reason for this is still unknown. Many have long envisioned a one-two punch approach to the treatment of blood cancer. The Global Car T Cell Therapy Market is estimated to be valued at US$ 1,085.0 million in 2021 and is expected to exhibit a CAGR of 24.5% during the forecast period (2021-2028). Chimeric antigen receptor (CAR) T-cell therapy uses a patient's own immune cells to fight the disease. It works by altering the genes on T cells in the body, which are normally responsible for killing virus and cancer-infected cells. This therapy is currently available only to people with certain types of blood cancer. However, it remains a promising option. The research into this new therapy is ongoing. The treatment is highly aggressive and is associated with potentially severe adverse events (AEs). Till date, no specific treatment for CAR T-cell therapy has been approved for the treatment of these two disorders. While most AEs are mild, some of them can be life-threatening. Since CAR T-cell therapy works on lymphoma and leukemia cells, it has been used to treat other types of cancer. Researchers have seen promising results, including in multiple myeloma and CLL. Further research is underway to find out how CAR T-cell therapy might be used in solid tumors. In the meantime, researchers are investigating CAR T-cell therapy's potential to treat solid tumors. Yescarta, an FDA-approved treatment for certain types of lymphoma, uses a patient's own white blood cells (T-cells) to fight cancer. The T-cells are removed from the patient by a similar process to blood donation and sent to Kite's specialized manufacturing facilities. The T-cells are genetically engineered to target the patient's cancer and expanded before being returned to the patient's body. Yescarta demonstrated clinically and statistically significant improvements in event-free survival (EFS), which is a measure of time since randomization until the first date of disease progression, the start of new therapy, or death. Although CAR T cell therapy is still relatively new, it is highly promising. The drug, tisagenlecleucel, is approved by the FDA to treat leukemia and certain forms of cancer in children and young adults. However, the side effects of this new treatment are serious, including life-threatening neurological events and cytokine-release syndrome, in order to reduce this risk, a drug named tocilizumab, has been approved by the Food and Drug Administration, for people with leukemia and other cancers. CAR T cell therapy is a medical procedure in which doctors inject engineered T cells into a patient's bloodstream to combat cancer. After chemotherapy, the body's immune system weakens and the new cells' task is to combat the cancer. These engineered T cells are able to fight the cancer because they're better suited to fight it. The procedure is typically an outpatient procedure, but some hospitals offer inpatient facilities as well. The patient must stay in an area near the hospital for at least four weeks, and they must be monitored closely. A multidisciplinary team should meet to discuss potential patients for the CAR T cell treatment. The team must take into account a patient's medical history, as well as physical health, to determine if the patient is suitable for the treatment. The requirements for CAR T cell therapy treatment are detailed in Table 1 and the EBMT guidelines. The minimum screening tests needed for CAR T cell therapy are also included in the list. Frozen Processed Food; Food Products Preserved Under Low Temperature and Used Over A Long Period7/14/2022 Processed food includes food that has been cooked, canned, frozen, packaged, and/or changed in nutritional composition with fortifying, preserving, or preparing in different ways. Freezing preserves food from the time it is prepared to the time it is eaten. Frozen food processing is the process of altering cooked food or ingredients and freezing it at recommended temperatures. They are easy to prepare as compared to fresh food and hence are gaining popularity across the globe. Frozen processed food control the microbial growth and retain the quality of processed products over long period. These products are stored under certain temperatures to maintain their quality. Frozen processed food is considered as healthy and natural food product.
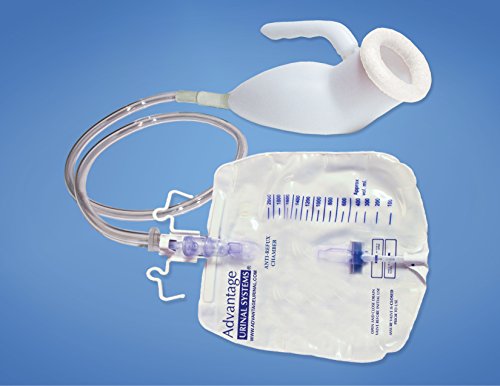
The Frozen Processed Food Market was valued at US$ 60.12 billion in 2016 and is expected to expand at a CAGR of 4.0%, in terms of revenue, over the forecast period (2017 – 2025). The frozen processed food industry is one of the fastest growing industries in the U.K. This is mainly due to the ever increasing demands from the people for nutritious, cost effective, and ready to eat food. Frozen processed food products are preserved under certain temperature and used over a long period. Food processing involves transformation of raw agricultural products into processed food, or from one type of processed food to another type. Processed food is used to replace the natural diet that people have and is made in huge quantities for profit. Processed food also accounts for much of the weight loss and diet control efforts of people who want to lose weight and are trying to eat healthier. Frozen processed food is defined as food products that are preserved under low temperature and used over a long period. Frozen foods retain their minerals and vitamins and there is no change in protein, carbohydrate, and/or fat content. Frozen foods, in some cases, contain more minerals and vitamins compared to fresh foods, as they lose minerals and vitamins over time while freezing preserves the nutrients. Food product properties of interest when considering freezing include thermal conductivity, specific heat, enthalpy, density, and latent heat. Frozen food is food that has been processed and stored without cooking. While there are many great benefits to eating frozen food, it does come with some risks and disadvantages as well. Some frozen foods contain large amounts of trans fats, which can not only clog the arteries but also make the user prone to heart diseases. Frozen food may also increases the cholesterol level of the body, which in turn leads to many heart related problems. Thus, it is important to know what the benefits and drawbacks are. Although, frozen foods are marketed to be healthy and nutritious but the fact is they can harmful for the body. Processed food has a bad reputation as a diet saboteur. It's blamed for obesity rates, high blood pressure and the rise of Type 2 diabetes. Moreover, frozen food has a long shelf-life; however, due to lack of infrastructure, the product is wasted easily leading to the loss for frozen food manufacturers. Freezing is an effective way to keep it fresh from the time it is prepared until the time the food is served. The freezing process itself does not destroy nutrients, there is little change in nutrient value during freezer storage. Ultra-processed foods tend to taste good and often inexpensive. However, they usually contain ingredients that could be harmful if consumed in excess, such as saturated fats, added sugar, and salt. It's important to change the urine collection bags as directed by the health care provider. Changing the bags too frequently can disrupt the closed drainage system in the urinary catheter. Expert opinion and manufacturer guidelines recommend changing the bags every five to seven days. There is no scientific evidence to support changing the bag more frequently, however. When changing the urine collection bags, the provider should also clean the connection to the catheter with alcohol wipes. If they can't get these items at a pharmacy, they can buy body-worn urine collection bags.
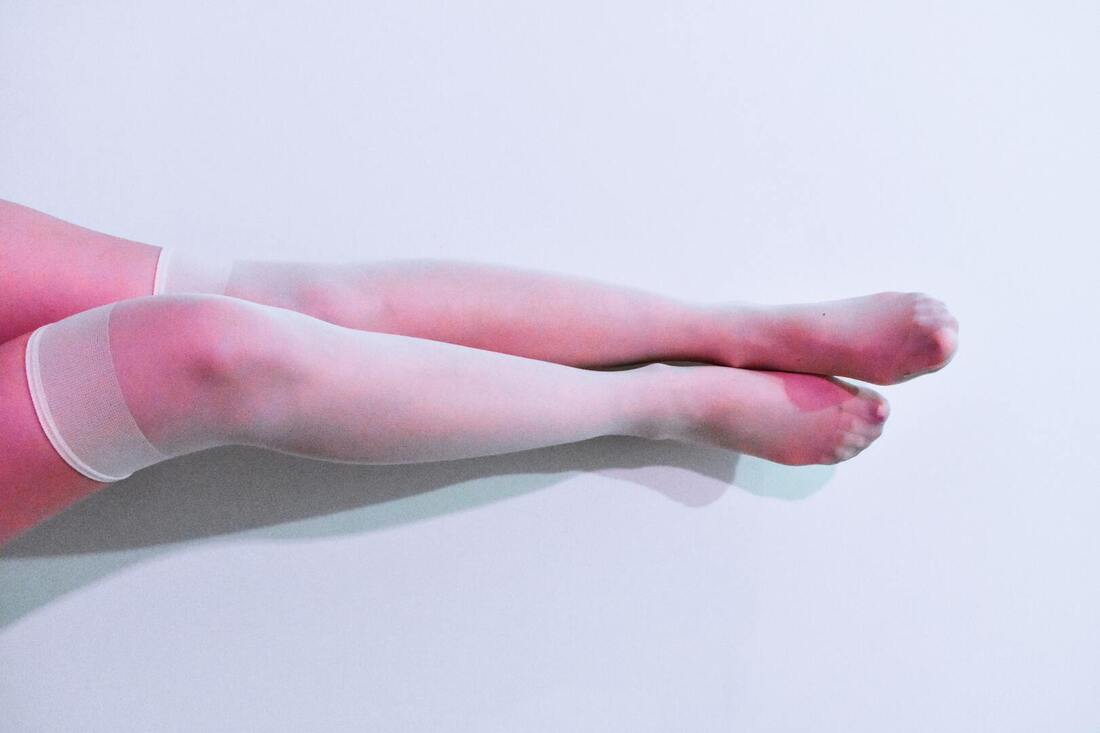
According to Coherent Market Insights the Urine Collection Bags Market Global Industry Insights, Trends, Outlook, and Opportunity Analysis, 2022-2028. In addition to these features, urine collection bags may be equipped with a braking interface tube 48. The braking interface tube material may be made of resilient thermoplastics, polyethylene, silicones, or co extrusions of these materials. PNC is a preferred material for the braking interface tube and is available in USP Class VI medical grade. When purchasing urine collection bags, providers should make sure that they're designed for the patient's use. One-way drainage is critical, but the collection bag and the connection tubing can also cause problems. Be sure to use a non-return valve on the urine collection bags in order to avoid spillage. Using this type of bag will prevent bacteria from reentering the urine and causing an infection. Urine collection bags with this feature has a non-return valve to prevent urine from returning along the catheter. Urinary leg bags can be worn under clothing and provide patients with discretion. The leg bags are often attached to a foley catheter, which channels urine while the patient recovers from spinal cord injury. They can choose between a non-ambulatory or ambulatory collection bags based on their needs. Consider the leg bag's capacity and usability, drainage valves, and straps before purchasing. These bags may be helpful for the health care provider. If the provider is helping the patient using a female urine collection bag, they may want to try a U-Bag. The U-Bag is a plastic collection bag with an oval opening. It's easy to attach and detach and is also sticky. Use it to collect a specimen. If patients are not comfortable putting the collection bag on themselves, they need to make sure to consult the health care provider before attempting this procedure. It's important to follow all precautions and instructions before starting a urine collection. Urine collection bags are typically made from two opposed pieces of flexible sheet material that are sealed at the seams to create a substantially closed receptacle for urine. In some cases, the seams are even heat-sealed or ultrasonic-welded. The urine collection bag may include a catheter. Before the catheter is inserted into the patient, the catheter extends into the urine collection bag. During this procedure, the catheter will pass urine from the bladder into the urine collection bag. Some patients find the leg bag uncomfortable, but it can be easily emptied. Some leg bags have a sleeve for drainage or a clamp that can be opened. It's important not to touch the tip of the drainage valve or the bladder. Providers need to Make sure to wash their hands afterward and write down how much urine the bag collected. This is necessary to ensure avoiding infections. Infection can be caused by a contaminated leg bag. Compression stockings are specialized hosiery that prevents and guards against venous disorders. These garments are elastic and wrapped around the leg. They work to compress the leg and reduce pressure on the blood vessels.
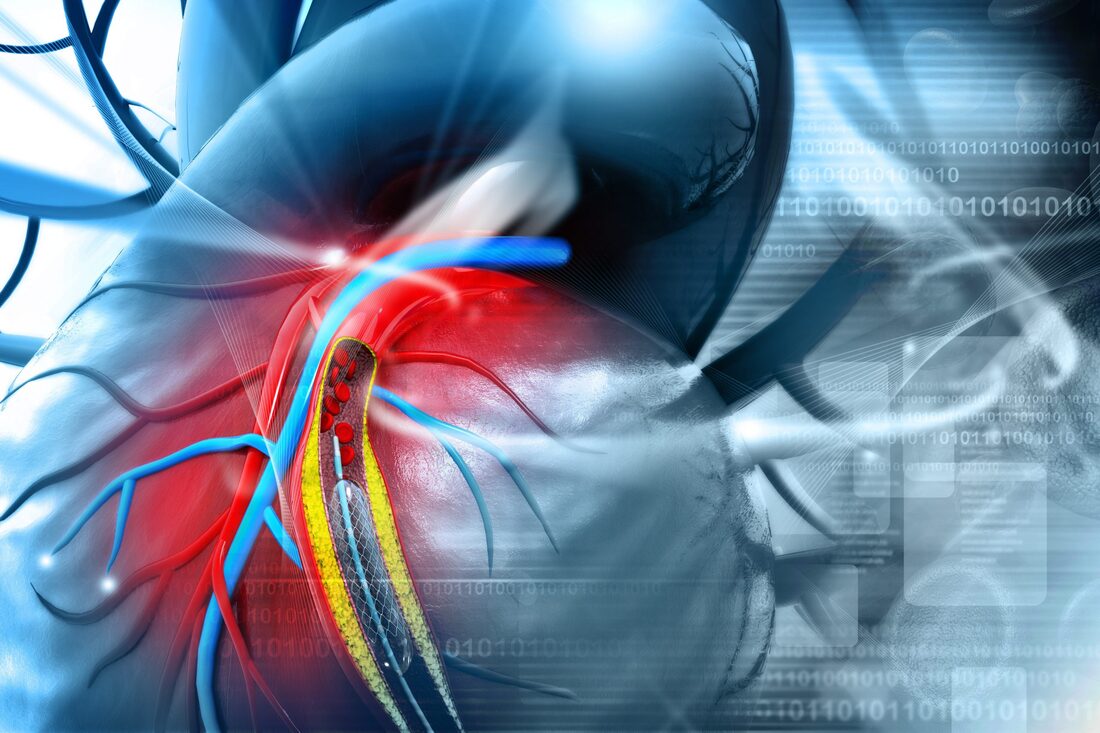
Compression stockings help increase blood flow to the legs. There are different strengths of compression stockings, which can be measured in millimeters of mercury. The lowest strength is eight to fifteen mmHg, which is usually sold over-the-counter. Hospitals often use these stockings to minimize the risk of blood clots. The strongest stockings are forty to fifty mmHg, which are commonly used for people with chronic venous insufficiency and lymphedema. According to Coherent Market Insights the Compression Stockings Market Global Industry Insight, Trends, Outlook and Opportunity Analysis, 2022-2028. Compression stockings are often prescribed by a doctor to help with medical conditions like varicose veins and spider veins. They also ease swelling in the ankles and legs. Some patients find them useful in preventing light-headedness and dizziness when standing for long periods of time. The socks also keep blood moving in the legs, which keeps it from pooling and forming clots, which can cause swelling and discoloration of the skin. The prevention of phlebitis with compression socks is a simple yet effective method of managing pain and swelling in the legs and arms. They are made of medical grade fabric and can be worn for up to two weeks at a time. They improve circulation and reduce swelling. Using compression stockings on the affected area will improve blood flow and alleviate pain. It is important to wear compression stockings as often as possible in order to prevent further complications. Phlebitis can be superficial or deep. The first type involves veins that are near the surface of the skin. This is the most common form and is typically self-limiting. Deep phlebitis can be life-threatening if the blood clot travels to the lungs. Compression stockings are a common treatment for varicose veins. Physicians usually recommend compression stockings as the first step in treating varicose veins, and they are also important after other procedures such as sclerotherapy and endovenous laser treatments. Varicose veins are visible in the legs, and can affect the look of the skin. Untreated, they can cause leg swelling and skin changes. In some cases, they can even cause blood clots to form, making the leg look uncomfortable. Treatment for varicose veins should begin as early as possible, before the condition worsens. After wearing compression stockings for an extended period of time, the condition may disappear completely. Nonmedical support hosiery are compression stockings that don't require a prescription and are intended to provide relief for tired and aching legs. Unlike prescription compression stockings, nonmedical support hosiery are designed to apply less pressure than prescribed socks. Compression stockings are available in different lengths, including knee-high, thigh-high, and waist-high varieties. They can apply different levels of compression, ranging from mild to firm. Compression socks apply pressure to the legs to help increase blood flow. If blood pools in the leg veins, it can cause clotting or damage. Compression socks are made of synthetic materials, and typically fall below the knee but may be worn up to the thigh as tights. Full-body compression garments are also available. Nonmedical support hosiery includes flight socks, elastic support socks, and other garments with compression throughout the garment. Patients who require graduated compression on their compression stockings should have a lower risk of recurrent venous insufficiency. Higher levels of compression can help reduce the risk of post-thrombotic syndrome (PTS) and another DVT. These conditions can also be known as chronic venous insufficiency or post-phlebitic syndrome. Gradient compression stockings can also help reduce swelling and pain from post-thrombotic syndrome. One type of graduated compression stockings is Jobst(r) Medical LegWear. These stockings come in three different compression levels - fifteen to twenty millimeters of mercury (mmHg), and are available in several styles. Using these stockings is a standard treatment for chronic venous insufficiency. Despite the success of graduated compression, it is important to note that not all patients tolerate these stockings well. Many sufferers report problems with compliance and discomfort while wearing the stockings. A cardiac stent can be used for treating a blocked coronary artery. These stents can also help in improving flow of blood after a heart attack. Cardiac stents are made up of metal mesh.
Stenting has proven to have a positive impact on many individuals, stenting along with angioplasty can help in saving life of an individual, if performed after a heart attack. Stents help in improving flow of blood and also prevents any further damage that can be caused to the heart muscles. These cardiovascular stents aids in improving symptoms of various heart diseases like chest pain and shortness of breath. Stenting also eliminates the need of coronary bypass surgery, as stenting is a less invasive procedure as compared to bypass surgery and the recovery time required in stenting is also less. It requires only a few days for recovering from stenting and it takes more than six weeks for recovering from bypass surgery. According to Coherent Market Insights the Cardiovascular Stents Market Global Industry Insights, Trends, Size, Share, Outlook, and Opportunity Analysis, 2022-2028. One of the main goals of cardiac stent development is to induce endothelialisation, a process believed to be key to long-term biocompatibility. Numerous strategies have been used to achieve this, including the use of porous biomaterials to promote spontaneous endothelial ingrowth. Furthermore, local delivery of pro-angiogenic growth factors has proven to induce transmural angiogenesis. Drug-eluting stents have made a significant impact in interventional cardiology. Compared to BMS, target TLR rates are down to 5% and the procedure has an overall success rate of 92.2%. DES have been proven effective in treating heart attacks and the latest studies have shown that fewer patients need revascularisation. However, there are some cautions when using this procedure. A recent meta-analysis showed that biodegradable polymer stents were not associated with lower ST rates compared with bare metal stents. The results, however, could be attributed to heterogeneity among the studies and differences in the types of non-biodegradable stents used. Furthermore, the majority of the studies used 1st or 2nd generation DES. Because of these limitations, findings from the meta-analysis cannot be given a high weight. Future studies should compare biodegradable polymers against non-biodegradable stents and evaluate whether or not the biodegradable stents are more effective for preventing or treating cardiovascular disease. The development of metal cardiovascular stents has benefited from the collaborative development of different materials, including new polymers and alloys. Researchers from various fields have worked together to develop ideal novel alloys, design more reasonable stent structures, and conduct long-term clinical trials to assess biocompatibility. However, these advancements have not yet been able to overcome the long-term problems associated with coronary thrombosis, such as the need to undergo a second procedure for a new stent. The coating on polyurethane cardiovascular stents is an excellent choice for biodegradable stents. In addition to its excellent biocompatibility, polyurethane has excellent mechanical properties. Its variety of monomers offers many advantages. In this review, we will describe the advantages of polyurethane cardiovascular stents. We will also discuss the pros and cons of polyurethane as a cardiovascular stent material. During the implantation process, an intentionally fractured stent may pose significant complications. A fractured stent can cause distortion and displacement of stent segments, and may impair the stent's function and embolization potential. Intentional stent fractures have occurred in a wide variety of cardiovascular stenting procedures, including coronary angioplasty. There are several advantages and disadvantages of biodegradable cardiovascular stents. These stents may require repeated interventions, but their benefits are not yet clear. For these reasons, further research is needed to determine whether biodegradable stents can provide optimal medical care. For example, they must be able to provide mechanical support to the arterial wall and be easily absorbed into the body. Breast Reconstruction is an operation to reconstruct or remake the breast after the mastectomy. Often, the reconstruction occurs after various operations. There are several Breast Reconstruction procedures. Many utilize the silicone or the saline breast grafts. Few other procedures utilize a flap of the tissue from the body. Breast Reconstruction can take place right after the breast carcinoma or it can take place after many months or years. One may have the operation to reconstruct the breasts or the patient may replace and change only one breast and rebuild it to make the other one similar to it. A patient may be advised for several operations over through various stages and phases. Few people opt to have the Breast Reconstruction after the lumpectomy however majority of them do not have. The decision or the choice to have the Breast Reconstruction is very secured private matter that cannot be disclosed. There are two main forms of the Breast Reconstruction operation after the lumpectomy. The flap reconstruction is one of the method in which the doctor takes the cells from the body and utilizes the same body cells to form or make a breast. Generally, they need a tissue from the lower belly. However, it can also be derived from the thigh, back and the bottom. The hospital providers call this a cell flap.
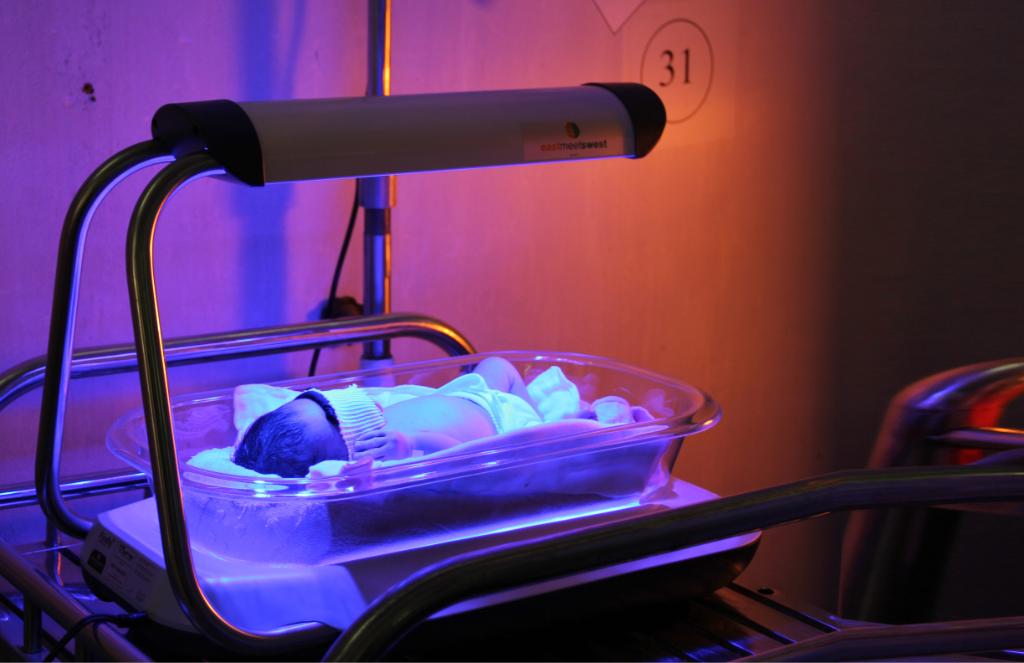
According to Coherent Market Insights the Breast Reconstruction Market Size, Share, Outlook, and Opportunity Analysis, 2022-2028. Generally, the healthcare experts shift a flap from the body and this process the flap holds its own plasma production or they may even detach the flap from its plasma distribution and then associate it to the blood vessels in the chest. There are several types of the flap reconstruction they are DIEP Flap, Tram Flap and the LD Flap. In the DIEP Flap the surgeon takes the fat, the dermis and the blood vessels from the lower abdomen and a DIEP flap do not eliminate the underlying layer of the belly. The surgeon removes the fat, blood vessels and the skin from the lower belly in the TRAM flap. The surgeons takes out the tissues and the thaw from the back of the body. They change and replace the LD Flap from the back to the chest portion or the region of the breast. IGAP Flap, is the process in which the tissue are taken from the butt and the muscles of the body aren’t utilized in this procedure. The SGAP Flap is a procedure which also eliminates the cells without any muscles for the butt and it utilizes a varied group of the blood vessels than the IGAP Flap technique.PAP Flap eliminates the tissues from the inner and the backside of the thigh and then it is employed to form a breast this method do not graft or alter the muscles from the thigh. The implant reconstruction is a method utilize by the doctors to remake the breast cells. Few doctors utilize a combination of the grafts and the cells from the body. Implant reconstruction can take place with the lumpectomy or one can do this after the process of the lumpectomy. Phototherapy equipment are often used to treat various skin-related diseases such as psoriasis, vitiligo, and others. Phototherapy, also known as light therapy, involves exposing the skin to ultraviolet light under medical supervision. Narrow-band UVB (NBUVB), PUVA (psoralen + UVA), Ultraviolet A (UVA), and UVA & NBUVB (in combination) are the common types of phototherapy used. However, the choice of the type of phototherapy equipment is dependent on the type of skin disease/condition. Light therapy, or phototherapy, is a method recognized by scientific medicine for the treatment of various diseases.
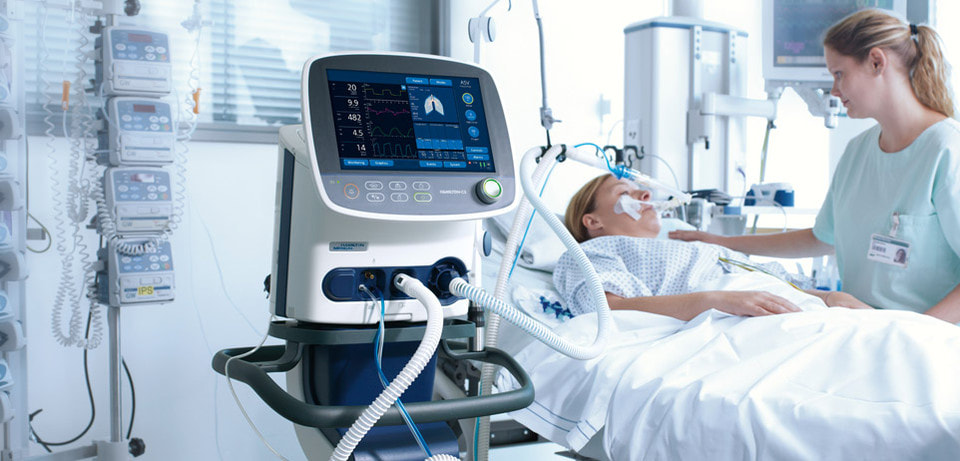
Light therapy, or phototherapy, with ultraviolet (UV) light effectively treats some types of skin diseases. The light source is used to mimic natural sunlight and hence assist in the treatment. The standard output of a light box is in range 2,500 and 10,000 lux. There are two basic types of phototherapy equipment. One type uses ultraviolet light, while the other consists of a combination of both wavelengths. Both are helpful for skin-related problems such as vitiligo, psoriasis, acne, and generalized itching. In addition to above-mentioned types, there are also home versions of phototherapy equipment. According to Coherent Market Insights the Phototherapy Equipment Market Global Industry Insights, Trends, Outlook, and Opportunity Analysis, 2022-2028. The home versions are typically more efficient because they allow the patient to go straight from the shower or bath to the lights. The effectiveness of medical phototherapy depends on the wavelength of the light, the area of the patient and its intensity. The phototherapy equipment uses state-of-the-art technology to provide high light output and long life. Infant phototherapy equipment can also help with the treatment of a newborn. It is also very compact, so it will be easy to transport and store. Its compact design save space, and does not produce excessive heat. Compared to other forms of therapy, light therapy is considered safe. Narrow-band UVB is the most common form of phototherapy used for the treatment of various skin diseases. NB-UVB is a type of light therapy used in the treatment of certain skin conditions such as chronic dermatitis, mycosis fungoides, psoriasis, and others. NB-UVB light treatments are given 2-3 times per week in the office. It is the most common form of phototherapy used to treat skin diseases. Narrowband refers to a specific wavelength of ultraviolet (UV) radiation, 311 to 312 nm. Narrowband UVB has since become the type of phototherapy most frequently used for the treatment of psoriasis and a wide range of skin disease. PUVA is a combination treatment which consists of Psoralens (P) and then exposing the skin to UVA (long wave UV radiation). The psoralen is used to treat psoriasis along with exposures to UV light in the UVA spectrum. PUVA is also of benefit in treating vitiligo, graft versus host disease, and mycosis fungoides (cutaneous T-cell lymphoma). Psoralens boost the amount of ultraviolet light the skin absorbs. This lets the light into the skin. The ultraviolet radiation helps treat severe skin diseases such as polymorphic light eruption, vitiligo, psoriasis, and cutaneous T-cell lymphoma, a type of cancer, among others. NB-UVB is most widely opted owing to its versatility and effectiveness for treating psoriasis, eczema, and mycosis fungoides patients. Phototherapy equipment includes LED phototherapy equipment, fiber-optic phototherapy equipment, and conventional phototherapy equipment. The mechanical ventilator is an artificial respiratory system that supplies oxygenation and ventilation to a patient. The device has a closed-loop control system, which allows the ventilation controller to automatically regulate its outputs. The ventilator sends out data about the patient's blood gas values and respiratory mechanics to an input data analyzer. The ventilator then regulates the amount of air being delivered to the patient to minimize respiratory work. The ventilator also measures the patient's heart rate, tidal volume, and inspiratory and expiratory times.
The main purpose of a mechanical ventilator is to allow the patient to recover from a respiratory condition by taking deep breaths and supporting their respiratory system. The ventilation is usually delivered through a respiratory circuit. There are three types of respiratory circuits. They are sometimes interchangeable. One type delivers positive pressure to the patient's airway, while the other two deliver negative pressure. A pulmonary function test is done to determine the correct respiratory circuit. The Global Mechanical Ventilators Market is estimated to account for US$ 4,248.7 Mn in terms of value by the end of 2027. The initial mode of mechanical ventilation is known as assist-control. As the name suggests, this mode ensures a minimum respiratory rate regardless of spontaneous breathing. The pressure-volume curve provides a relationship between the volume and the pressure, so any given volume corresponds to a specific amount of pressure. The resultant airway pressure is not fixed but varies depending on the resistance, elastance, and flow rate of the respiratory system. Although mechanical ventilators are primarily used in hospitals and emergency situations, they can also be used at home to provide supplemental oxygen for long-term respiratory illnesses. However, home care caregivers must have adequate resources to support their patients while on a mechanical ventilator. Those in need of mechanical ventilation should also be aware that this treatment can increase the risk of developing pneumonia and damaging the vocal cords. So, use of mechanical ventilator is very important for respiratory related disease. Intentional leaks can interfere with the ventilator's pressurization. Intentional mask leaks may prevent VT from being achieved, but pressure ventilatory support compensates for these effects. Most ventilators now include a tidal volume guaranteed (TVG) mode, labelled as "dual mode ventilation." To achieve VTG, technician need to increase the flow and the pressure applied to the airway. PEEP (positive airway pressure) is another mode of therapy that can be used with mechanical ventilators. Increasing intrathoracic pressure increases the end-expired lung volume. The increased airspace at the end of expiration prevents atelectasis. Higher levels of PEEP may improve oxygenation in some pulmonary disorders and in some cases limit cardiogenic pulmonary edema. It also limits lung injury caused by high FIO2. The intrinsic PEEP can be measured in a passive patient with the use of an end-expiratory hold maneuver. The pressure at the end of expiration reflects the alveolar pressure. However, it is important to note that neuromuscular blockade is not advisable to be used for intrinsic PEEP measurement. Expiratory flow tracing is another method for determining intrinsic PEEP. The latter is often more reliable than the former. |


 RSS Feed
RSS Feed
